CSP-270 cream contains a novel selective glucocorticoid receptor agonist (GW870086) in development for the treatment of Dry Eye Disease due to Meibomian Gland Dysfunction (MGD). Uniquely the proprietary cream is applied once daily to the outside of the eyelid where it forms a depot that delivers drug to the surface of the eye every time the patient blinks.
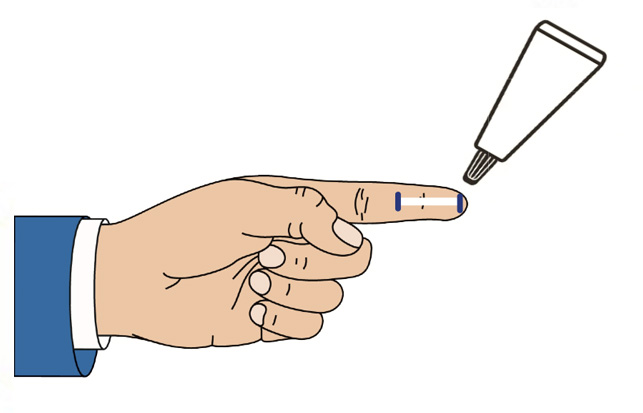
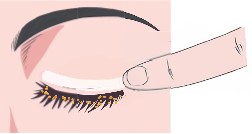
This novel route of administration offers competitive advantages over other treatments for dry eye disease, increasing the concentration of drug delivered to ocular structures and reducing the risk of wash-out due to tearing when compared toeye drops.
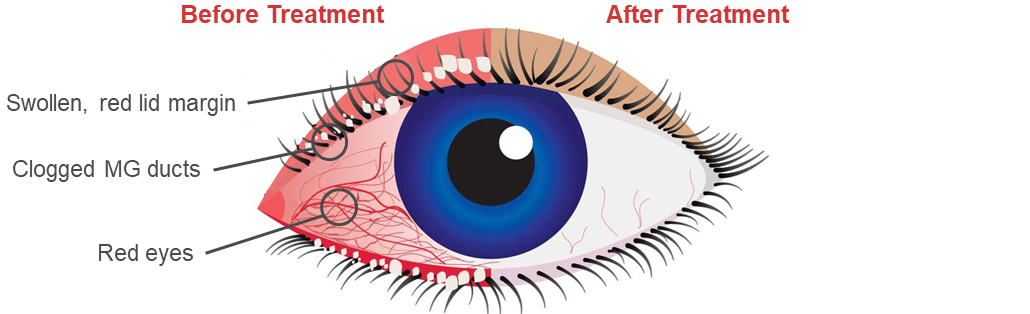
CSP-270 has achieved clinical proof-of-concept in patients with dry eye disease (DED) due to MGD. In phase 2 clinical studies, CSP-270 showed a 30% improvement in VAS of ocular discomfort after 3 weeks treatment. The product, which is phase 3 ready, is safe and well tolerated with no ocular TEAES and no increases in intraocular pressure (IOP) reported.
The prevalence of DED is expected to grow dramatically owing largely to an ageing population and increasing use of digital screens. There is high unmet need for a treatment with a faster onset of action. Convenience of dosing, rapid onset of action and lack of IOP increase are major strengths of the CSP-270 cream, competitive strengths that have been estimated to translate to peak revenues of between $230M and $630M by 2035 in the US alone
Partnership Opportunities
CSP acquired global rights to CSP-270 following the acquisition of AxeroVision [press release] and is planning to commercialise the product in Greater China. CSP is actively seeking partners to co-fund the additional development and commercialization in major territories such as the USA, Europe and Japan.
For further details please contact: simon.bennett@cspharmaceuticals.com
